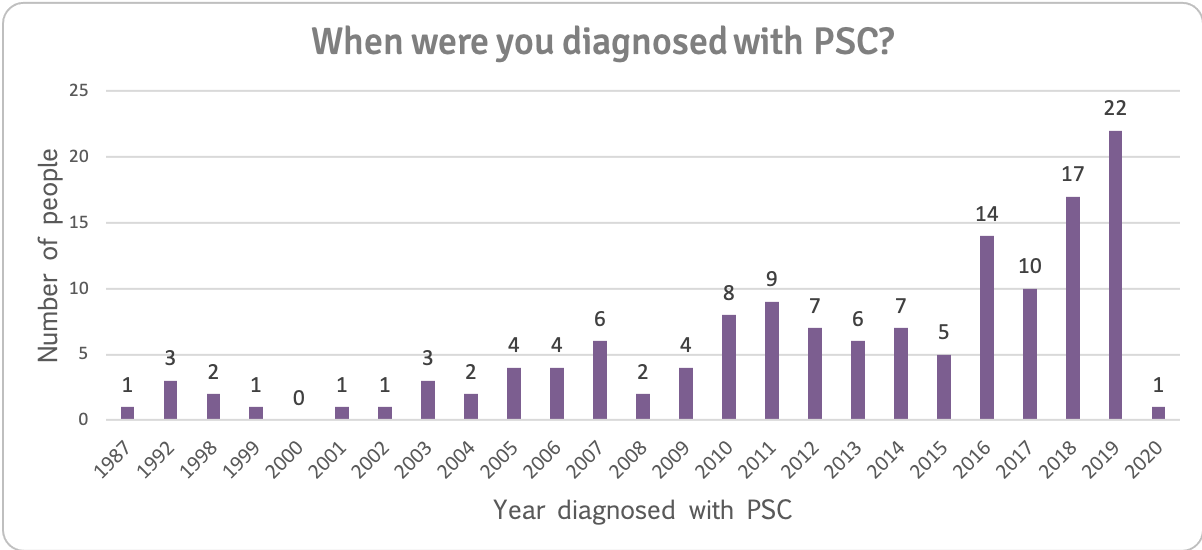
Interest in taking part
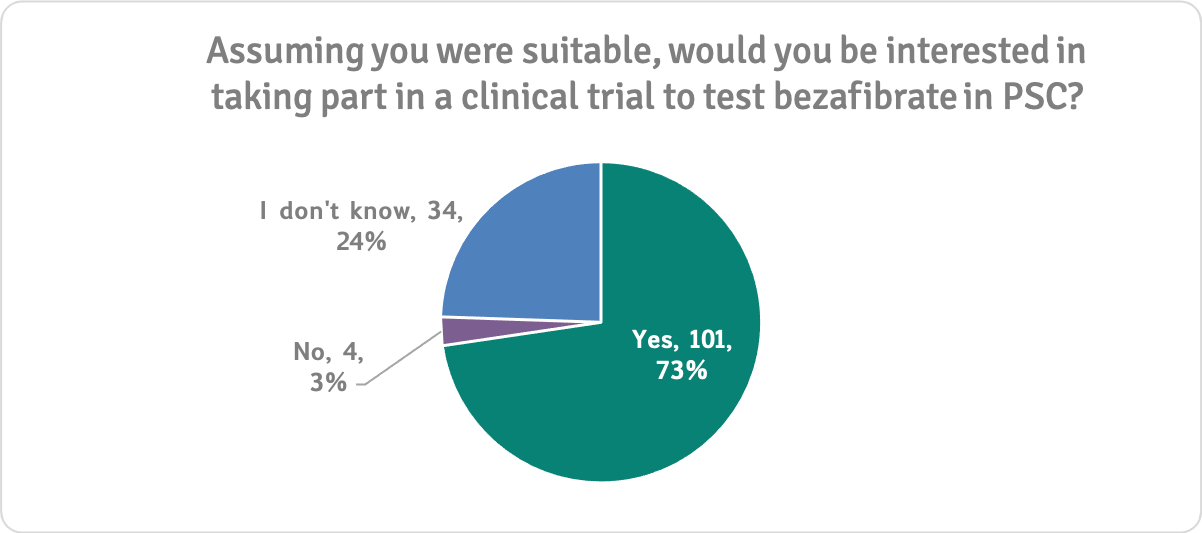
Most people (79%) were interested in taking part in research to test bezafibrates in PSC. This corroborates our previous survey findings and is understandable, given that people who have PSC are living with an incurable, progressive liver disease with an increased risk of aggressive cancers and sudden complications. They desperately need an effective treatment.
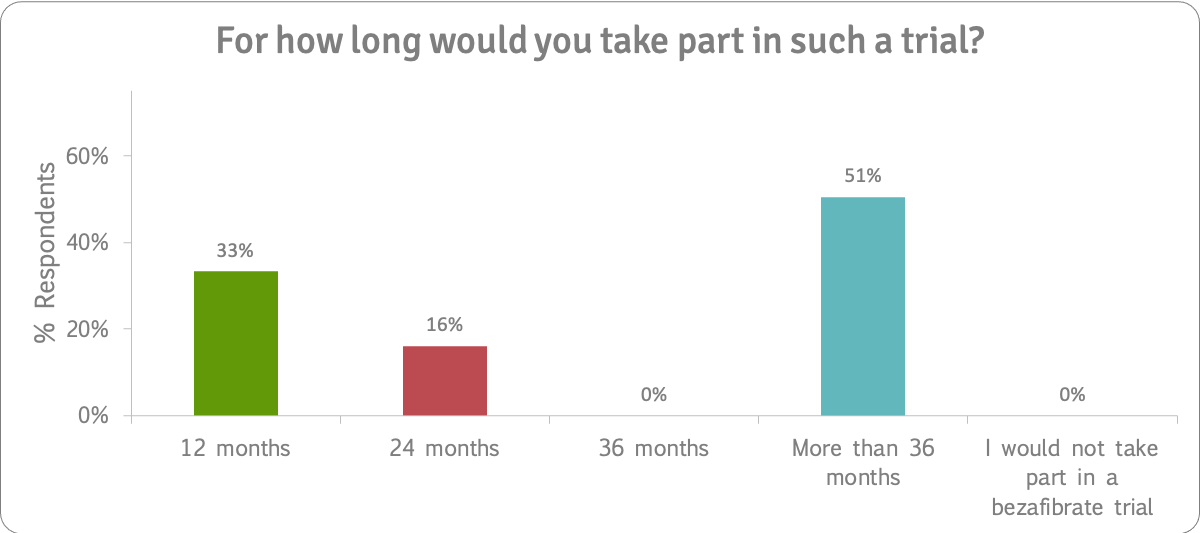
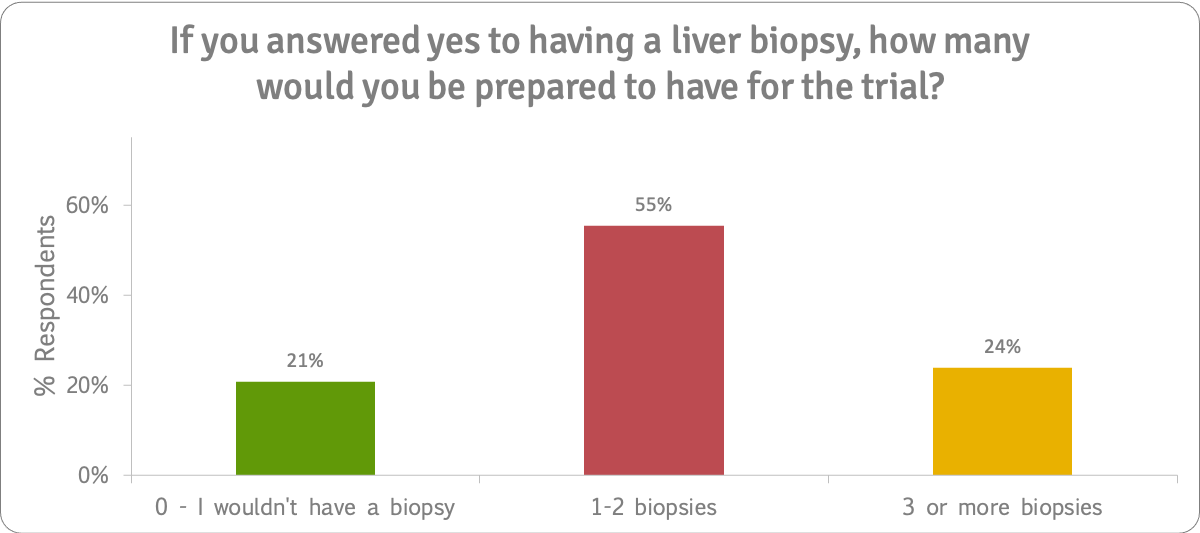
The survey results showed that not only were patients (51%) prepared to take part in a trial that lasted three years or more, they were prepared to have at least one biopsy (79%) in a bezafibrate trial. A quarter (24%) said they would be prepared to have three or more biopsies.
Our focus group discussed the issue of biopsies and agreed that if biopsies were used in clinical trials, potential participants should be told why the information biopsies give is so crucial.
Removing barriers to recruitment
PSC is a rare disease and researchers are mindful that recruitment could be an issue, that is, finding enough patients to take part.
When we looked at the reasons people gave for not being sure about taking part, or not being interested at all, we found that the reasons were centred around factors that could be modified by investigators, such as providing comprehensive information about the study drug and trial design. PSC frequently affects people in their thirties and forties, that is, people who are working and raising young families, often full time, and study design information allows them to make decisions about the feasibility of participation according to their own circumstances.
Patients place great value on the opinion of their consultants. Efforts should be made to educate the healthcare professionals that care for patients, or use study sites affiliated with the UK-PSC biobank network, where healthcare professionals are familiar with PSC and current research.
Meaningful endpoints
What makes a treatment effective? What is important to people affected by PSC? We asked an open-ended question on what should be recorded or measured in this trial in addition to blood tests, symptoms and safety/side effects. 61 people gave comments.
People focused on measuring the effect of bezafibrate on disease progression (both in the liver and bile ducts) and PSC complications. They also wanted to know what effect the drug would have on co-existing diseases (especially inflammatory bowel disease) and cancer risk. Quality of life, wellbeing and symptoms (in particular, itch) were also a priority for patients. Some people mentioned extending blood tests to include vitamin levels and cholesterol. Finally, some mentioned hard endpoints: long term survival and need for liver transplant.
High quality trial design
When patients take part in clinical trials, they do so in order to make a difference to themselves and/or to others. Clinical trials must have a high-quality design to give robust results that can be relied upon. People with PSC do not have the luxury of time to wait for trials of long duration using hard endpoints (time to transplant or death).
PSC can lead to liver failure, cancers, reduced quality of life and numerous unplanned hospital admissions. People with PSC are classed as 'vulnerable' by the UK government, and 'extremely vulnerable' if they have had a transplant in relation to COVID-19. A transplants may be life-saving, but it means a lifetime of immunosuppression. In the current COVID-19 outbreak, this is frightening. People with PSC need a treatment to slow or halt progression of the disease with upmost urgency.