CM-101
FULLY RECRUITED
The SPRING Study
Thinking of taking part?
- Check how suitable you are by looking at the information below.
- Check our map to see if you can get to a study site.
- Contact the research team for more details if you think you are suitable. Check back regularly as we are adding more contact info every day.
- Got a question? Our Clinical Trials Pack has lots of information about how to join clinical trials, switching hospitals for trials and what's involved.
- Contact us if you need any help.
Quick Links
Update on Eligibility Criteria - 01 September 2023
The eligibility criteria have widened to include people who are:
- on a stable dose of biologics for IBD
- taking vancomycin
About CM-101
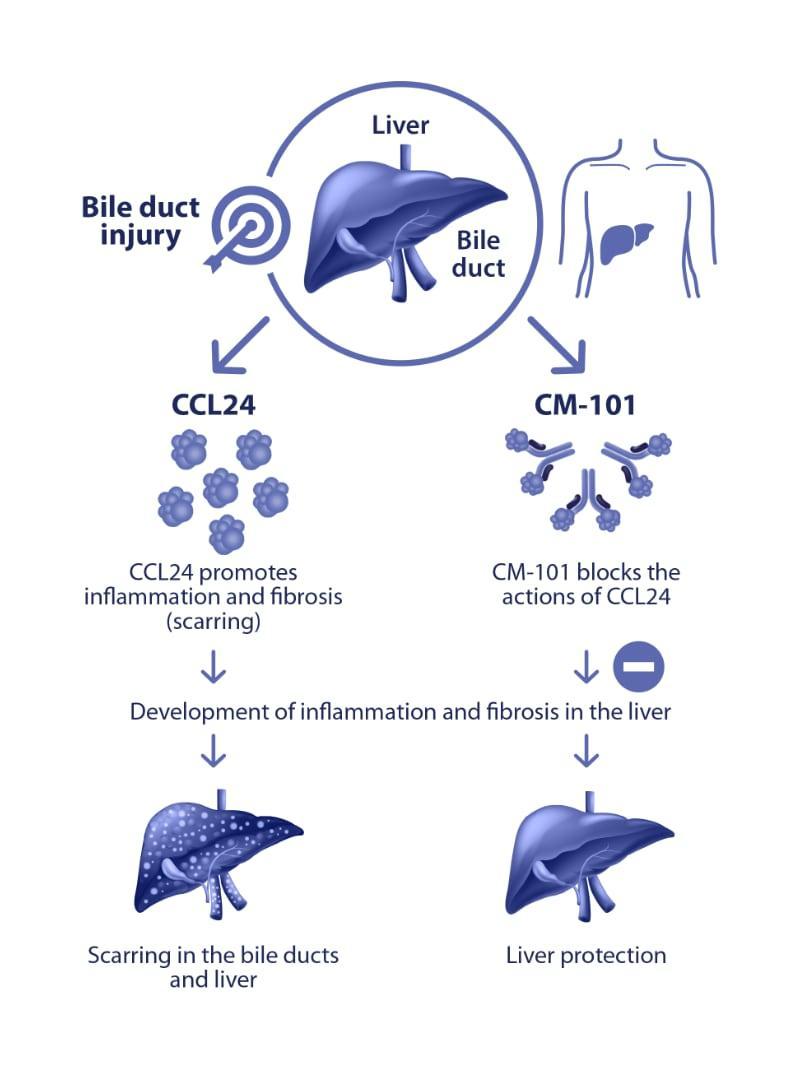
CCL24 is a small protein involved in cell “communication” that helps regulate inflammation and is involved in the development of fibrosis (scarring). CCL24 is found in higher concentrations in PSC patients than the general population.
CM-101 is a monoclonal antibody, created by Chemomab, that can block the actions of CCL24 and thus its role in the development of inflammation and fibrosis. CM-101 was tested in several animal models, including the MDR2 knockout mice (mice that have severe fibrosis in the bile ducts and liver, eventually developing cirrhosis). These tests showed that CM-101 significantly reduced liver fibrosis, and markers that indicate the presence of inflammation in the bile ducts.
Chemomab's video, available to watch here, explains how the drug is thought to work.
FAQs
| Trial Name | The SPRING Study
A Phase 2a, Randomized, Double-Blind, Placebo-Controlled, Clinical Trial Evaluating The Safety And Efficacy Of CM-101 In Subjects With Primary Sclerosing Cholangitis- The Spring Study |
| ClinicalTrials.gov Identifier | NCT04595825 |
| Phase | Phase 2a trial |
| Intervention | CM-101 intravenous infusions or Placebo (inactive substance) over an hour - once every three weeks for 5 administrations.
After completion of the 5 administrations, there is an option to receive CM-101 intravenous infusion over an hour – once every three weeks for an additional 11 administrations. |
| Recruiting now? | No - this study has recruited all participants for this phase of the research |
| Do I qualify to take part? | Find out more from PatientWing |
| Participating Centres | See PSC Support map, the current sites list on Clinicaltrials.gov and UK-PSC for up-to-date status for each centre. |
| Liver disease diagnosis | You must have a diagnosis of large duct PSC (combination of large and small duct PSC is allowed).
Note autoimmune hepatitis is allowed as long as it isn’t considered to be more active than PSC. |
| Age | You must be an adult (18 years and above). |
| Females: can I be pregnant? | No |
| Is IBD allowed? | Yes - if it is mild and in remission - no colitis flare within the previous 90 days. |
| Can I continue to take biologics for my IBD? | Yes - if dose of the biologics is stable for at least 12 weeks before screening. |
| Is there an ALP (alkaline phosphatase) requirement? | ALP should be at least 1.5 times the upper limit of the normal range (ULN), during screening. |
| Can I take UDCA (Urso)? | Yes - UDCA is allowed if you are on a stable dose (dose must not exceed 23mg/kg/day) and have been taking it for at least 12 weeks. |
| Can I take vancomycin? | Yes - Vancomycin as treatment for PSC is allowed if on a stable for at least 12 weeks before screening. |
| Can I take part if I have a stent? | No |
| Are recent acute cholangitis (ie bacterial cholangitis) attacks allowed? | No acute cholangitis attack within 90 days of randomisation. |
| Can I take part if I have had a liver transplant? | No |
| Is cirrhosis allowed? | No |
| Previous studies and information |
|
| Are travel expenses covered? | Reasonable refreshments, meals, travel expenses and associated other out of pocket expenses (e.g. parking) will be reimbursed.
If an overnight stay is needed, it will be reimbursed. |
| Procedures and tests include: | Blood samples, Fibroscan, ultrasound scans, urine sample (+pregnancy testing), patient questionnaires. |
| How many study visits will there be? How long will each study visit be? | The study includes a double-blind* treatment period, once every 3 weeks for 12 weeks.
After completion of the double-blind treatment period, you will have the option to enter the Open-Label Treatment Period**. You will receive CM-101 intravenous infusion every 3 weeks for 33 weeks. After the last infusion, there will be a 15 weeks of safety follow-up including 2 visits and 1 phone call. (All treatment visits will take at least 3 hours) *double-blind means that neither you nor the researcher is told whether you are taking the treatment (and at what dose) or placebo. **Open-label treatment period means that you will have the option to take the actual treatment after taking part in the double-blind treatment period. |
| Duration of study | Double-blind treatment period of 15 weeks
Open-label treatment period of 33 weeks. (You will have the option to take the actual treatment after taking part in the double-blind treatment period) Follow-up period of 15 weeks following last treatment visit |
| Basic design of study | Randomised, Double Blind, Placebo Controlled Study
Volunteers will be randomised to receive one of two doses of the study drug (CM-101 10 mg/kg or 20mg/kg ) or placebo. You will have an 80% chance of getting the study drug. The dose you receive will depend on when you enter the study. You will have a 20% chance of getting the placebo. If you agree to participate in the Open-label portion you will receive the active dose of which ever cohort you were randomised to in the double blind portion (10 mg/kg or 20 mg/kg) |
| How is this intervention thought to work and what is it thought to do? | In PSC, it is thought in imbalance of some substances in the body causes various kind of cells to move to the liver, causing inflammation and fibrosis (scarring), leading to liver damage. CM-101 is a drug in development that is believed to be able to block the activity of these substances and thus limiting the liver damage.
Chemomab's video, available to watch here, explains how the drug is thought to work. |
| Link to Sponsor | Chemomab |