Vancomycin
The PSC-Vanc Study - NO LONGER RECRUITING
Thinking of taking part?
- Check how suitable you are by looking at the information below.
- Check our map to see if you can get to a study site.
- Contact the research team for more details if you think you are suitable. Check back regularly as we are adding more contact info every day.
- Got a question? Our Clinical Trials Pack has lots of information about how to join clinical trials, switching hospitals for trials and what's involved.
- Contact us if you need any help.
Quick Links
THIS TRIAL HAS NOW RECRUITED ALL PARTICIPANTS
Primary sclerosing cholangitis (PSC) is a chronic liver disease of an unknown cause and with no effective medical treatment. Nearly three-quarters of people living with PSC also have a form of inflammatory bowel disease (PSC-IBD).
People with PSC-IBD appear to be at greater risk of colon cancer and the need for bowel surgery than people who don’t also have PSC.
Dr Nabil Quraishi (University of Birmingham, UK) recently discovered that the bacteria found in the bowel in people with PSC-IBD were very different to those with ulcerative colitis alone and healthy individuals. These bacteria had genes that were associated with converting bile acids into a form that can be absorbed into the colonic tissue. Analyses of genes obtained from biopsies of the colon from people with PSC-IBD suggested that the pathways that control the levels of bile acid in the colon were in a state of overdrive
These new findings raised several important questions in helping us understand how PSC and PSC-IBD develop and if these differences in the gut bacteria have anything to do with it.
Find out about clinical trials
Vancomycin
Interestingly, an oral antibiotic called oral vancomycin has been shown to improve both liver blood tests and bowel inflammation in people with PSC-IBD. This antibiotic does not get absorbed so is its mode of action is possibly due to a change in the gut bacteria.
The PSC-Vanc Study
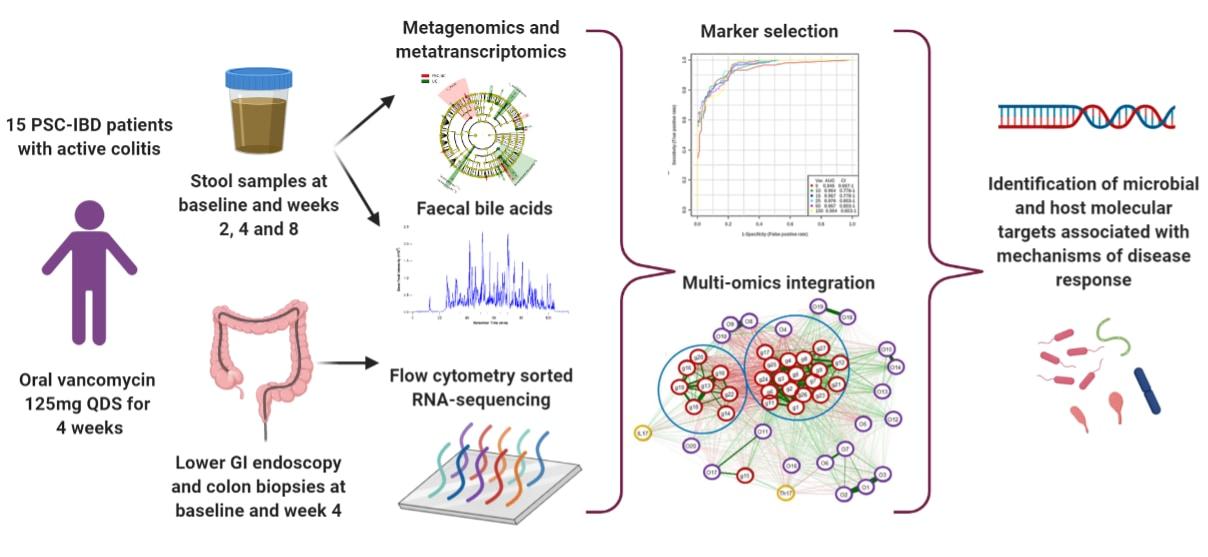
Dr Quraishi will lead a new study looking at vancomycin in PSC patients called the PSC-Vanc Study. The study team will recruit fifteen people with PSC-IBD who are having their annual colonoscopy. If they find inflammation in the bowel, those participants will be given 4 weeks’ of treatment with oral vancomycin. They will collect stool samples and colonic biopsies at different intervals. The team will then evaluate changes in the gut bacteria, the chemicals produced, bile acids and genes being expressed within the bowel. This will help us to identify specific disease pathways that are associated with PSC-IBD.
Why is this PSC study important?
Dr Quraishi said, “We don't know why vancomycin seems to work in PSC and PSC-IBD. The big question is are we doing something to the gut bacteria or bile acids that lead to a favourable shift. If so what is it and why? It’s a simple study but an important study to do. We are hoping to unlock and understand potential mechanisms for colitis and liver disease in PSC and if there are specific targets (such as bacteria, bile acids and genes) that can be translated into novel treatments for clinical trials.”
Key information
| Trial Name |
A systems biology approach for identification of host and microbial mechanisms and druggable targets for the treatment of PSC-IBD (PSC-Vanc) |
| Phase | Observational study |
| Intervention | Oral vancomycin |
| ClinicalTrials.gov Identifier | NCT05376228 |
| Recruiting now? | No, fully recruited |
| Participating Centres and Contact (if possible) | University Hospitals Birmingham NHS Foundation Trust
Contact Dr Quraishi (nabil.quraishi@nhs.net) to take part. |
| Liver disease diagnosis | PSC (pre or post transplant) |
| Age | 18 years or over |
| Females: can I be pregnant? | No |
| Is IBD allowed? | Yes |
| Is there an ALP (alkaline phosphatase) requirement? | No |
| Can I take UDCA (Urso)? | Yes |
| Can I take part if I have a stent? | Yes |
| Are recent acute cholangitis attacks allowed? | Yes as long as no antibiotics given in the last three months |
| Can I take part if I have had a liver transplant? | Yes |
| Is cirrhosis allowed? | Yes as long as no evidence of liver decompensation |
| Previous studies and information | Quraishi MN, Acharjee A, Beggs AD, Horniblow R, Tselepis C, Gkoutos G, Ghosh S, Rossiter AE, Loman N, van Schaik W, Withers D, Walters JRF, Hirschfield GM, Iqbal TH. A Pilot Integrative Analysis of Colonic Gene Expression, Gut Microbiota, and Immune Infiltration in Primary Sclerosing Cholangitis-Inflammatory Bowel Disease: Association of Disease With Bile Acid Pathways. J Crohns Colitis. 2020 Jul 30;14(7):935-947. doi: 10.1093/ecco-jcc/jjaa021. PMID: 32016358; PMCID: PMC7392170. |
| Travel expenses? | Not included at present |
| Procedures and tests include: | Surveillance colonoscopy at baseline (if due), followed by a limited flexible sigmoidoscopy 4 weeks later (after completion of oral vancomycin course) |
| How many Study Visits? How long will each study visit be? | At least two face to face visits timed with colonoscopy / flexible sigmoidoscopy with the remaining three visits being virtual (over the phone). |
| Duration of Study (treatment duration/follow-up) | 4 weeks of oral vancomycin with a further follow up for 4 weeks (total 8 weeks duration) |
| Basic design of study eg placebo etc | Single arm study: oral vancomycin as open label active treatment arm with no comparator group. |
| Link to Patient Information (or pdf) | Download Patient Information Sheet |
| Link to Sponsor | University Hospitals Birmingham |